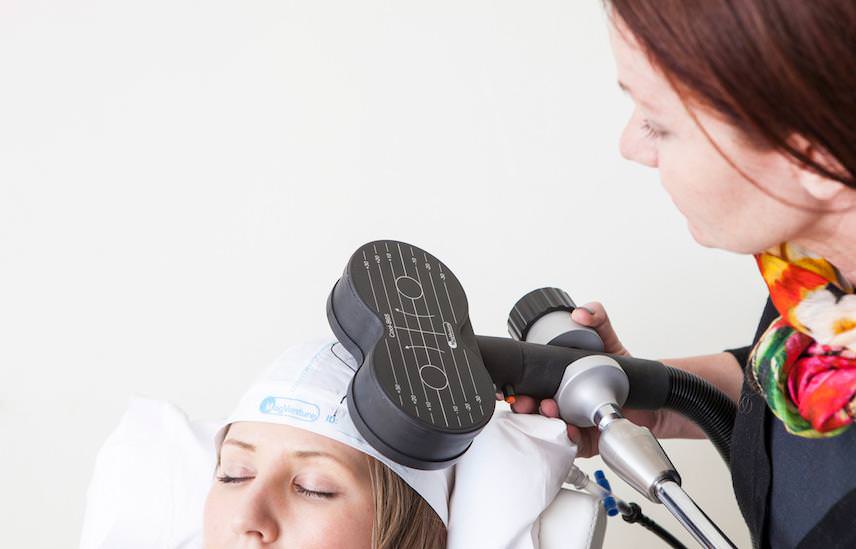
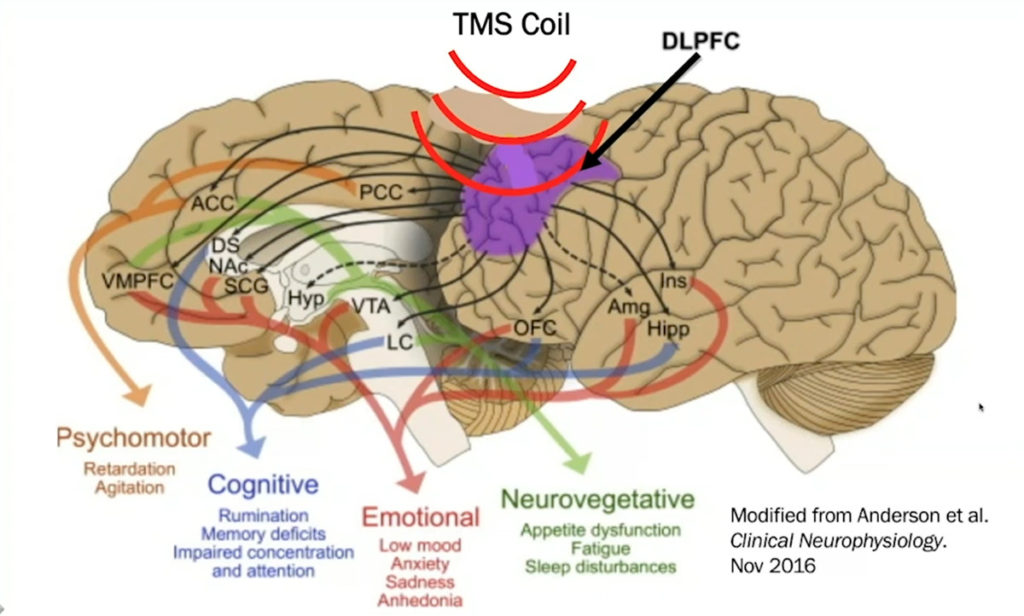
Repetitive Transcranial Magnetic Stimulation (rTMS)
- Depression is real and it starts in your brain.
So why aren’t you treating your brain … with Transcranial Magnetic Stimulation? - For just 3 minutes a day
- No need of hospitalization.
So you can regain your quality of life. - Approved therapy by American and European organisations (FDA, NICE, CE)
- No need for drugs
- Non-invasive
- Comfortable Setting
- Advanced Technology
- Convenient Location
- Highly trained medical staff.
Welcome to ΙΨΚ rTMS clinic.
Stop tolerating depression, treat it!